Prebiotic Spotlight: Global Prebiotic Association’s Standards of Evidence for Prebiotic Classification
In this edition of the Global Prebiotic Association’s (GPA) Prebiotic Spotlight, the focus is on GPA’s Standards of Evidence (SoE), a tiered framework of criteria designed to classify prebiotic candidates based on the strength and quality of the scientific evidence supporting their prebiotic activity and associated health effects.
Overview
The term “prebiotic” has evolved significantly since its inception three decades ago. Currently, the industry continues to face challenges in establishing a globally unified regulatory standard to define and categorize prebiotics. These challenges are further compounded by the complexity of the microbiome, the emergence of new and novel ingredients with potential prebiotic properties, and inconsistencies in study designs and research methodologies employed across the field.
To address these challenges, GPA has led internal discussions with representatives from both the industry and academia to develop a SoE framework. This framework classifies prebiotic candidates into three categories: Established, Novel, and Emerging, based on the robustness of the available scientific evidence. As such, this report examines the resulting tiered system and its potential role in advancing the field toward more rigorous clinical validation and greater scientific standardization.
Evolution of the Prebiotic Criteria
The scientific understanding of prebiotics has evolved from simple, gut-focused, fiber-based definitions to more sophisticated recognition of microbial utilization and functional effects.
A prebiotic is defined by GPA as “a compound or ingredient that is utilized by the microbiota producing a health or performance benefit” (Deehan et al., 2024). This updated definition broadens the scope beyond traditional dietary fibers to include non-fiber substances such as polyphenols and omega-3 fatty acids, and recognizes microbiome niches beyond the gut, including skin, respiratory system, and reproductive system.
GPA also introduced a specific definition for the “prebiotic effect,” describing it as “a health or performance benefit that arises from alteration of the composition and/or activity of the microbiota, as a direct or indirect result of the utilization of a specific and well-defined compound or ingredient by microorganisms”.
Beyond the designation of an ingredient as a prebiotic based on demonstrated activity and resulting effects, ingredients may be further categorized into one of three levels recognized within the scientific community, depending on the extent of available research. This classification is primarily determined by whether an ingredient has demonstrated both microbiota modulation and measurable health benefits in human subjects. These two criteria represent the core requirements for prebiotic designation, supported by clinical substantiation, with preference given to studies demonstrating both outcomes concomitantly.
Comparative Evidence Frameworks: GPA and ISAPP
GPA’s SoE initiative builds upon the foundational work in the prebiotic field, including that of the International Scientific Association for Probiotics and Prebiotics (ISAPP).
In alignment with ISAPP’s criteria outlined by Hutkins et al. (2025), GPA recognizes the importance of a prebiotic candidate having a well-defined chemical structure, along with established safety and purity, supported by both preclinical evaluation and clinical validation.
Furthermore, both organizations emphasize that a prebiotic candidate must demonstrate microbiota modulation and health benefits, with GPA acknowledging this as the gold standard. GPA also allows for differentiated levels of substantiation across categories, with “Novel” and “Emerging” designations permitting separate substantiation studies for their support.
GPA’s Standards of Evidence
GPA classifies prebiotic candidates into three categories based on the volume and quality of available human clinical data supporting them.
- Established: This category requires robust human randomized controlled trials (RCTs), with demonstrated repeatability and a simultaneous correlation between microbiota modulation and health benefits within the same study. Currently recognized examples include inulin, acacia gum, fructooligosaccharides, galactooligosaccharides, human milk oligosaccharides, and resistant starches (types 2 & 3).
- Novel: This category requires at minimum one human RCT demonstrating simultaneous correlation, or multiple separate studies substantiating microbiota modulation and health benefits. Examples include relatively more recently characterized candidates with growing evidence, such as certain polyphenols and omega-3s.
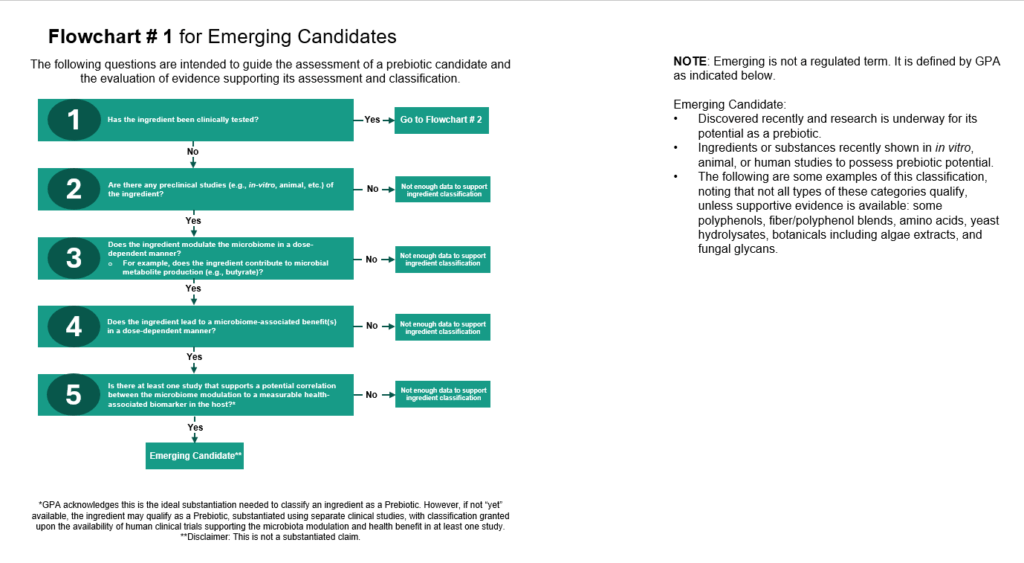
- Emerging: This category is primarily supported by in vitro or animal data, with human research underway. More than one study is required that demonstrate a potential correlation to a measurable health biomarker. Examples currently include fungal glycans, botanicals, amino acids, and yeast hydrolysates.
To reiterate, while these terms are not formally regulated, they are widely accepted and used within the scientific community and industry.
Clinical and Regulatory Implications
Implementing GPA’s tiered SoE system provides a standardized framework for prebiotic innovation, that enhances both clinical transparency and regulatory compliance within the prebiotic industry. By categorizing ingredients as Established, Novel, or Emerging, the system
allows manufacturers to align marketing language with the specific depth of supporting clinical evidence, effectively reducing the risk of “prebiotic washing” and unsubstantiated health claims. For researchers, this structure offers a clear roadmap for study design, with an emphasis on dose-dependent responses and measurable correlations between microbiota modulation and host benefits.
Ultimately, this tiered approach supports greater consumer trust by providing a verifiable pathway for prebiotic validation, including certification programs like NutrastrongTM Prebiotic Verified, which certifies ingredients as well as finished products containing prebiotics that meet defined quality standards and scientific evidence. At the same time, GPA’s SoE provides regulatory bodies with a scientifically rigorous benchmark to evaluate the efficacy and safety of diverse prebiotic substrates across global markets.
Conclusions and Future Directions
GPA’s SoE framework represents an advancement in the scientific structuring and evaluation of the prebiotic category. By establishing clear criteria for “Established”, “Novel”, and “Emerging” ingredients, GPA has created an interactive and easy-to-follow format that assists industry professionals navigate complex regulatory landscapes.
The objective of establishing and implementing this standard is to reduce discrepancies in study design and methodology approaches, while providing a structured pathway for ingredients to be reclassified into higher tiers as new, robust, and confirmatory clinical evidence becomes available.
Looking ahead, it is recommended that future clinical trials focus on evaluating the prebiotic effect of newly identified and categorized ingredients in well-defined populations to better characterize their health and performance benefits. In addition, research is expected to expand beyond the gut to examine other microbiome mechanisms affecting the entire body, as well as to explore novel health and performance benefits, including cognitive and physical performance endpoints. Furthermore, dose specificity remains an important consideration for maintaining the integrity of the prebiotic classification as new ingredients are considered.
Finally, while designed as a self-directed resource, these tiered standards are intended to enhance both public and regulatory trust in the development of prebiotic ingredients and finished products, ensuring that the designation “prebiotic” remains anchored in rigorously substantiated health and performance outcomes.
References:
Deehan, E. C., Al Antwan, S., Witwer, R. S., Guerra, P., John, T., & Monheit, L. (2024). Revisiting the Concepts of Prebiotic and Prebiotic Effect in Light of Scientific and Regulatory Progress-A Consensus Paper from the Global Prebiotic Association. Advances in nutrition (Bethesda, Md.), 15(12), 100329. https://doi.org/10.1016/j.advnut.2024.100329
Hutkins, R., Walter, J., Gibson, G. R., Bedu-Ferrari, C., Scott, K., Tancredi, D. J., Wijeyesekera, A., & Sanders, M. E. (2025). Classifying compounds as prebiotics – scientific perspectives and recommendations. Nature reviews. Gastroenterology & hepatology, 22(1), 54–70. https://doi.org/10.1038/s41575-024-00981-6